|
ChemicalBook Optimization Suppliers |
|
| 化学名: | 2,3-プロパンジオール | | 英語化学名: | Propylene glycol | | 别名: | PROPYLENEGLYCOL, REAGENT (ACS)PROPYLENEGLYCOL, REAGENT (ACS)PROPYLENEGLYCOL, REAGENT (ACS);Propyledne glycol;Glycerol Impurity 3(Glycerol EP Impurity C);Propylene glycol 57-55-6;Propyleneglycol Manufacturer;1,2-PROPANEDIOL, GR ACS 99.5%;PROPYLENEGLYCOLTECHGRADE;Propylene Glycol, Reagent | | CAS番号: | 57-55-6 | | 分子式: | C3H8O2 | | 分子量: | 76.09 | | EINECS: | 200-338-0 | | カテゴリ情報: | Medicine grade;INORGANIC & ORGANIC CHEMICALS;57-55-6 | | Mol File: | 57-55-6.mol | 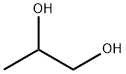 |
| 融点 | -60 °C (lit.) | | 沸点 | 187 °C (lit.) | | 比重(密度) | 1.036 g/mL at 25 °C (lit.) | | 蒸気密度 | 2.62 (vs air) | | 蒸気圧 | 0.08 mm Hg ( 20 °C) | | 屈折率 | n20/D 1.432(lit.) | | FEMA | 2940 | PROPYLENE GLYCOL | | 闪点 | 225 °F | | 貯蔵温度 | Store at +5°C to +30°C. | | 溶解性 | Chloroform (Slightly), Ethyl Acetate (Slightly), Methanol (Slightly) | | 酸解離定数(Pka) | 14.49±0.20(Predicted) | | 外見 | Viscous Liquid | | 色 | APHA: ≤10 | | 比重 | 1.038 (20/20℃)1.036~1.040 | | 臭い (Odor) | at 100.00 %. odorless very slight alcoholic | | PH | 6-8 (100g/l, H2O, 20℃) | | 爆発限界(explosive limit) | 2.4-17.4%(V) | | においのタイプ | odorless | | 水溶解度 | miscible | | Sensitive | Hygroscopic | | Merck | 14,7855 | | JECFA Number | 925 | | BRN | 1340498 | | InChIKey | DNIAPMSPPWPWGF-UHFFFAOYSA-N | | LogP | -0.92 | | CAS データベース | 57-55-6(CAS DataBase Reference) | | NISTの化学物質情報 | Propylene glycol(57-55-6) | | EPAの化学物質情報 | Propylene glycol (57-55-6) |
| | 2,3-プロパンジオール Usage And Synthesis |
| 外観 | 無色澄明の液体 | | 定義 | PGはプロピレングリコールの表示名称である。本品は、次の化学式で表される二価アルコールである。 | | 溶解性 | 水, アルコール, エーテルに可溶。水及びエタノールに極めて溶けやすい。 | | 解説 | プロピレングリコール化学式 CH3CHOHCH2OH 。プロピレンクロロヒドリンを炭酸ナトリウムと水で処理して得られる無色粘稠な液体。沸点 188℃。溶剤,不凍液,殺菌剤としても用いられる。可塑剤やポリエステル樹脂の原料に使われ,医薬として水性膏剤,水に難溶の物質の注射用溶剤に用いられる。
| | 用途 | 合成樹脂、可塑剤、界面活性剤、溶剤、保存剤、湿潤剤。 | | 用途 | 食品加工剤(香料、色素の溶剤、保存剤、湿潤剤)。 | | 用途 | 有機合成原料、ポリエステル樹脂 | | 用途 | 繊維,樹脂,溶剤,界面活性剤,不凍液,食品加工,医薬品など多方面に用途がある。 | | 化粧品の成分用途 | 保湿.湿潤剤、保水剤、減粘剤、皮膚コンディショニング剤、溶剤、香料 | | 効能 | 湿潤剤, 溶解剤, 懸濁剤 | | 商品名 | プロピレングリコール (丸石製薬); プロピレングリコール (日興製薬); プロピレングリコール (日興製薬) | | 使用上の注意 | 吸湿性あり。 | | 説明 | Propylene glycol is used as antifreeze in breweries and
diaries, in the manufacture of resins, as a solvent, and
as an emulsifier in food. It was present as an
occupational sensitizer in the color-film developer
Flexicolor. | | 化学的特性 | Propylene glycol is a colorless, odorless,
syrupy liquid. | | 化学的特性 | Propylene glycol has a slight, characteristic taste. It is practically odorless. It absorbs moisture when exposed to moist
air. | | 天然物の起源 | Reported found in several varieties of mushrooms, roasted sesame seed, oat groats, parmesan cheese, cocoa,
pecans and truffle. | | 使用 | Propylene Glycol is a humectant and flavor solvent that is a polyhy-
dric alcohol (polyol). it is a clear, viscous liquid with complete solu-
bility in water at 20°c and good oil solvency. it functions as a
humectant, as do glycerol and sorbitol, in maintaining the desired
moisture content and texture in foods such as shredded coconut
and icings. it functions as a solvent for flavors and colors that are
insoluble in water. it is also used in beverages and candy. | | 使用 | Next to water, propylene glycol is the most common moisturecarrying vehicle used in cosmetic formulations. It has better skin permeation than glycerin, and it also gives a pleasant feel with less greasiness than glycerin. Propylene glycol is used as a humectant because it absorbs water from the air. It also serves as a solvent for anti-oxidants and preservatives. In addition, it has preservative properties against bacteria and fungi when used in concentrations of 16 percent or higher. There is a concern that propylene glycol is an irritant at high concentrations, though it appears to be quite safe at usage levels under 5 percent. | | 使用 | Used as a solvent. | | 調製方法 | Propylene glycol generally is synthesized commercially by
starting with propylene, converting to the chlorohydrin, and
hydrolyzing to propylene oxide, which is then hydrolyzed
to propylene glycol. It can also be prepared by other
methods. | | 定義 | An alcohol in which the hydroxyl groups are attached to a carbon atom of a branched or straight-chain aliphatic hydrocarbon. | | 製造方法 | Manufactured by treating propylene with chlorinated water to form the chlorohydrin, which is converted to the glycol by
treatment with sodium carbonate solution. It is also prepared by heating glycerol with sodium hydroxide. | | 適応症 | Propylene glycol solution (40% to 60%, v/vCH2CH[OH]CH2OH, propylene glycol)
applied to the skin under plastic occlusion hydrates the skin and causes desquamation
of scales. Propylene glycol, isotonic in 2% concentration, is a widely used vehicle
in dermatologic preparations. Hydroalcoholic gels containing propylene glycol or
other substances augment the keratolytic action of salicylic acid. Keralyt gel consists
of 6% salicylic acid, 19.4% alcohol, hydroxypropylcellulose, propylene glycol, and water and is an extremely effective keratolytic agent. Overnight occlusion is used
nightly until improvement is evident, at which time the frequency of therapy can
be decreased to every third night or once weekly. This therapy is well tolerated,
is usually nonirritating, and has been most successful in patients with X-linked
ichthyosis vulgaris. Burning and stinging may occur when applied to damaged skin.
Patients with other abnormalities of keratinization with hyperkeratosis, scaling, and
dryness may also benefit. | | brand name | Sentry Propylene Glycol (Union Carbide); Sirlene (Dow Chemical). | | Aroma threshold values | Detection: 340 ppm | | 一般的な説明 | Thick odorless colorless liquid. Mixes with water. | | 空気と水の反応 | Water soluble. | | 反応プロフィール | 1,2-Propanediol is hygroscopic. 1,2-Propanediol is sensitive to excessive heat (tends to oxidize at high temperatures). 1,2-Propanediol can react with oxidizing materials. 1,2-Propanediol is incompatible with acid chlorides, acid anhydrides, chloroformates, and reducing agents. 1,2-Propanediol dissolves many essential oils. A mixture of 1,2-Propanediol with hydrofluoric acid and silver nitrate was put in a glass bottle which burst 30 minutes later. | | 危険性 | Toxic. | | 健康ハザード | Liquid may irritate eyes. | | 火災危険 | 1,2-Propanediol is combustible. | | 接触アレルゲン | Propylene glycol is used as a solvent, a vehicle for
topical medicaments such as corticosteroids or aciclovir,
an emulsifier and humectant in food and cosmetics,
and as antifreeze in breweries, in the manufactures
of resins. It was present as an occupational sensitizer
in the color film developer Flexicolor?. Patch tests in
aqua are sometimes irritant. | | 安全性プロファイル | Slightly toxic by
ingestion, skin contact, intraperitoneal,
intravenous, subcutaneous, and
intramuscular routes. Human systemic
effects by ingestion: general anesthesia,
convulsions, changes in surface EEG.
Experimental teratogenic and reproductive
effects. An eye and human skin irritant.
Mutation data reported. Combustible liquid
when exposed to heat or flame; can react
with oxidizing materials. Explosive in the
form of vapor when exposed to heat or
flame. May react with hydrofluoric acid +
nitric acid + silver nitrate to form the
explosive silver fulminate. To fight fire, use
alcohol foam. When heated to
decomposition it emits acrid smoke and
irritating fumes. | | 職業ばく露 | Propylene glycol is used as a solvent;
emulsifying agent; food and feed additive; flavor, in manu-
facture of plastics; as a plasticizer, surface-active agent;
antifreeze, solvent, disinfectant, hydroscopic agent; coolant
in refrigeration systems; pharmaceutical, brake fluid; and
many others. | | 発がん性 | Dewhurst et al. and
Baldwin et al. in studies on the carcinogenicity of
other chemicals used propylene glycol as the solvent. As a
result they tested propylene glycol alone for carcinogenic
activity in rats and mice. Dewhurst et al. used a single
injection of 0.2 mL, whereas Baldwin et al. gave rats
and mice three to five subcutaneous injections, amount not
specified. In neither case were tumors observed during a
period of about a year or 2 years .
Wallenious and Lecholm applied propylene glycol
to the skin of rats three times a week for 14 months but found
no tumor formation. Stenback and Shubik confirmed
these findings when they applied propylene glycol at undiluted
strength and as a 50 and 10% solution in acetone to the
skin of mice during their lifetimes.
No tumors have been reported in the lifetime dietary
feeding studies . In fact, Gaunt et al.
specifically state that no tumors were found in the rats. | | 環境運命予測 | Propylene glycol can be released into the environment via
industrial releases or by disposal of consumer products.
Propylene glycol is readily soluble in water and has a low
sorption partition coefficient (KOC), so has the ability to move
through soil and to leach into ground water. Because of low
vapor pressure (0.1 mmHg at 25°�C) and high water solubility,
there is minimal volatilization to the atmosphere from
surface water releases as well as substantial removal of its
vapors by wet deposition. Its low octanol/water partition
coefficient (KOW) indicates that bioconcentration and biomagnification
should not happen. Propylene glycol is readily
degraded in surface water and soil, by chemical oxidation and
microbial digestion, with a short half-life (1–5 days) in
aerobic or anaerobic conditions. It is also rapidly degraded in
the atmosphere by photochemical oxidation, with a half-life
about 1 day. Although environmental releases can and do
occur (airports must monitor storm water runoff of deicing
solutions), human health effects are likely to be minor, at
least in comparison to effects from potential exposures in
clinical scenarios. | | 純化方法 | Dry the diol with Na2SO4, decant and distil it under reduced pressure. [Beilstein 1 IV 2468.] | | Toxicity evaluation | Propylene glycol has a low degree of toxicity in animals as well
as humans, such that very high doses are needed to elicit effects
in acute toxicity studies. The toxic effects of propylene glycol
appear to be similar in animals and in humans. Central
nervous system (CNS) depression, hematologic toxicity,
hyperosmolarity, metabolic acidosis, cardiovascular effects,
and renal toxicity encompass the main acute and subacute
syndromes for propylene glycol. Most of the effects of
propylene glycol can be ascribed to high concentrations of the
parent molecule or to the accumulation of D,L-lactate in the
blood. Due to its alcohol moiety, propylene glycol at very high
concentrations is the most likely reason for the CNS depression.
Also, because high concentrations of propylene glycol will
increase the osmolarity of the blood, the hyperosmotic effects
are likely due to the parent molecule. The cardiovascular and
renal effects may be a result of the hyperosmolarity in combination
with the metabolic acidosis. The acidosis itself results
from the accumulation of lactate (both D- and L-forms), which
has been well documented in both animals and humans. | | 不和合性 | Incompatible with oxidizers (chlorates,
nitrates, peroxides, permanganates, perchlorates, chlorine,
bromine, fluorine, etc.); contact may cause fires or explo-
sions. Keep away from alkaline materials, strong acids
(especially nitric acid), strong bases, permanganates,
dichromates; may cause a violent reaction. | | 廃棄物の処理 | Dissolve or mix the
material with a combustible solvent and burn in a chemical
incinerator equipped with an afterburner and scrubber.All federal, state, and local environmental regulations must
be observed. |
|