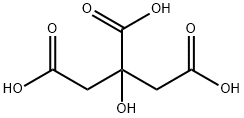
시트르산
|
|
시트르산 속성
- 녹는점
- 153-159 °C (lit.)
- 끓는 점
- 248.08°C (rough estimate)
- 밀도
- 1.67 g/cm3 at 20 °C
- 증기 밀도
- 7.26 (vs air)
- 증기압
- <0.1 hPa (20 °C)
- 굴절률
- 1.493~1.509
- FEMA
- 2306 | CITRIC ACID
- 인화점
- 100 °C
- 저장 조건
- 2-8°C
- 용해도
- Citric acid also dissolves in absolute (anhydrous) ethanol (76 parts of citric acid per 100 parts of ethanol) at 15 °C.
- 물리적 상태
- grit
- 산도 계수 (pKa)
- 3.14(at 20℃)
- 색상
- White
- 수소이온지수(pH)
- 3.24(1 mM solution);2.62(10 mM solution);2.08(100 mM solution);
- 냄새
- Odorless
- ?? ??
- odorless
- 폭발한계
- 8%, 65°F
- 수용성
- soluble in Water (1174g/L at 10°C, 1809g/L at 30°C, 3825g/L at 80°C).
- 감도
- Hygroscopic
- 최대 파장(λmax)
- λ: 260 nm Amax: 0.20
λ: 280 nm Amax: 0.10
- Merck
- 14,2326
- JECFA Number
- 218
- BRN
- 782061
- 안정성
- Stable. Incompatible with bases, strong oxidizing agents, reducing agents, metal nitrates.
- InChIKey
- KRKNYBCHXYNGOX-UHFFFAOYSA-N
- LogP
- -1.64
- CAS 데이터베이스
- 77-92-9(CAS DataBase Reference)
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | Xi,C,T | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 41-36/37/38-36/38-37/38-34-36-35-61-60 | ||
| 안전지침서 | 26-39-37/39-24/25-36/37/39-45-36-53 | ||
| 유엔번호(UN No.) | UN 1789 8/PG 3 | ||
| WGK 독일 | 1 | ||
| RTECS 번호 | GE7350000 | ||
| F 고인화성물질 | 9 | ||
| TSCA | Yes | ||
| HS 번호 | 2918 14 00 | ||
| 유해 물질 데이터 | 77-92-9(Hazardous Substances Data) | ||
| 독성 | LD50 in mice, rats (mmol/kg): 5.0, 4.6 i.p. (Gruber, Halbeisen) | ||
| 기존화학 물질 | KE-20831 |
시트르산 C화학적 특성, 용도, 생산
개요
Citric acid is a white, crystalline, weak organic acid present in most plants and many animals as an intermediate in cellular respiration. Citric acid contains three carboxyl groups making it a carboxylic, more specifically a tricarboxylic, acid.the name citrus originates from the Greek kedromelon meaning apple of melon for the fruit citron. Greek works mention kitron, kitrion, or kitreos for citron fruit, which is an oblong fruit several inches long from the scrublike tree Citrus medica. Lemons and limes have high citric acid content, which may account for up to 8% of the fruit's dry weight.
Citric acid is a weak acid and loses hydrogen ions from its three carboxyl groups (COOH) in solution.the loss of a hydrogen ion from each group in the molecule results in the citrate ion,C3H5O(COO)33. A citric acid molecule also forms intermediate ions when one or two hydrogen atoms in the carboxyl groups ionize.the citrate ion combines with metals to form salts, the most common of which is calcium citrate. Citric acid forms esters to produce various citrates, for example trimethyl citrate and triethyl citrate.
화학적 성질
Citric acid is a weak organic acid with the formula C6H8O7. It is a natural preservative / conservative and is also used to add an acidic, or sour, taste to foods and soft drinks. In biochemistry, the conjugate base of citric acid, citrate, is important as an intermediate in the citric acid cycle, which occurs in the metabolism of all aerobic organisms.Citric acid is a commodity chemical, and more than a million tonnes are produced every year by fermentation. It is used mainly as an acidifier, as a flavoring, and as a chelating agent.
물리적 성질
CITRIC ACID, white crystalline solid, decomposes at higher temperatures, sp gr 1.542. Citric acid is soluble in H2O or alcohol and slightly soluble in ether. The compound is a tribasic acid, forming mono-, di-, and tri- series of salts and esters.It occurs in large amounts is citrus fruits, and is used widely in industry as an acidifier, as a flavoring and chelating agent. pKa values are 5.21, 4.28 and 2.92 at 25 °C (extrapolated to zero ionic strength).Citric acid is a good buffering agent for solutions between about pH 2 and pH 8. It is popular in many buffers in many techniques, electrophoresis (SSC Buffer #), to stop reactions, for biopurifications, crystallography... In biological systems around pH 7, the two species present are the citrate ion and mono-hydrogen citrate ion. the pH of a 1 mM solution of citric acid will be about 3.2.
출처
Citric acid exists in greater than trace amounts in a variety of fruits and vegetables, most notably citrus fruits. Lemons and limes have particularly high concentrations of the acid; it can constitute as much as 8 % of the dry weight of these fruits (about 47 g/L in the juices ) . The concentrations of citric acid in citrus fruits range from 0.005 mol/L for oranges and grapefruits to 0.30 mol/L in lemons and limes. Within species, these values vary depending on the cultivar and the circumstances in which the fruit was grown.역사
The discovery of citric acid is credited to Jabir ibn Hayyan (Latin name Geber, 721–815).Citric acid was first isolated in 1784 by the Swedish chemist Carl Wilhelm Scheele (1742–1786), who crystallized it from lemon juice.
The crystalline structure of anhydrous citric acid, obtained by cooling hot concentrated solution of the monohydrate form, was first elucidated by Yuill and Bennett in 1934 by X-ray diffraction.
In 1960 Nordman and co-workers further suggested that in the anhydrous form two molecules of the acid are linked through hydrogen bonds between two –COOH groups of each monomer.
용도
Citric Acid is an acidulant and antioxidant produced by mold fermentation of sugar solutions and by extraction from lemon juice, lime juice, and pineapple canning residue. it is the predominant acid in oranges, lemons, and limes. it exists in anhydrous and monohydrate forms. the anhydrous form is crystallized in hot solutions and the monohydrate form is crystallized from cold (below 36.5°c) solutions. anhydrous citric acid has a solubility of 146 g and monohydrate citric acid has a solubility of 175 g/100 ml of distilled water at 20°c. a 1% solution has a ph of 2.3 at 25°c. it is a hygroscopic, strong acid of tart flavor. it is used as an acidulant in fruit drinks and carbonated beverages at 0.25-0.40%, in cheese at 3-4%, and in jellies. it is used as an antioxidant in instant potatoes, wheat chips, and potato sticks, where it prevents spoilage by trapping the metal ions. it is used in combination with antioxidants in the processing of fresh frozen fruits to prevent discoloration.주요 응용
Citric acid is a weak organic acid that is known as a commodity chemical, as more than a million tonnes are produced every year by mycological fermentation on an industrial scale using crude sugar sol utions, such as molasses and strains of Aspergillus niger. Citric acid is widely distributed in plants and in animal tissues and fluids and exist in greater than grace amounts in variety of fruits and vegetables, most notably in citrus fruits such as lemon and limes. Citric acid is mainly used as an acidifier, flavoring agent and chelating agent. It was also used as a chemical restrainer particularly in developers for the collodion process and in silver nitrate solutions used for sensitizing salted and albumen papers.제조 방법
By mycological fermentation using molasses and strains of Aspergillus niger; from citrus juices and pineapple wastes정의
ChEBI: Citric acid is a tricarboxylic acid that is propane-1,2,3-tricarboxylic acid bearing a hydroxy substituent at position 2. It is an important metabolite in the pathway of all aerobic organisms. It has a role as a food acidity regulator, a chelator, an antimicrobial agent and a fundamental metabolite. It is a conjugate acid of a citrate(1-) and a citrate anion.일반 설명
Citric acid appears as colorless, odorless crystals with an acid taste. Denser than water. (USCG, 1999)공기와 물의 반응
The pure material is moisture sensitive (undergoes slow hydrolysis) Water soluble.반응 프로필
Citric acid reacts with oxidizing agents, bases, reducing agents and metal nitrates . Reactions with metal nitrates are potentially explosive. Heating to the point of decomposition causes emission of acrid smoke and fumes [Lewis].건강위험
Inhalation of dust irritates nose and throat. Contact with eyes causes irritation.Biotechnological Applications
Citric acid cycleCitrate, the conjugate base of citric acid is one of a series of compounds involved in the physiological oxidation of fats, proteins, and carbohydrates to carbon dioxide and water.
This series of chemical reactions is central to nearly all metabolic reactions, and is the source of two-thirds of the foodderived energy in higher organisms. Hans Adolf Krebs received the 1953 Nobel Prize in Physiology or Medicine for the discovery. The series of reactions is known by various names, including the "citric acid cycle", the "Krebs cycle" or "Szent-Gy?rgyi — Krebs cycle", and the "tricarboxylic acid (TCA) cycle".
Other biological roles
Citrate is a critical component of bone, helping to regulate the size of calcium crystals.
Safety Profile
Poison by intravenous route. Moderately toxic by subcutaneous and intraperitoneal routes. Mildly toxic byingestion. A severe eye and moderate skin irritant. An irritating organic acid, some allergenic properties. Combustible liquid. Potentially explosive reaction with metal nitrates. When heated to decomposition it emits acrid smoke and fumes.시트르산 준비 용품 및 원자재
원자재
준비 용품
시트르산암모늄
TERT-BUTYL 4-(3-FORMYLBENZYL)TETRAHYDRO-1(2H)-PYRAZINECARBOXYLATE
아세틸 트라이뷰틸 시틀산염
METHYL 3-((PIPERIDIN-4-YL)METHYL)BENZOATE
N-isopropyl-2-(piperazin-1-yl)pyridin-3-amine
1-CYCLOPROPYLPIPERAZINE
4-((1-(TERT-BUTOXYCARBONYL)PIPERIDIN-4-YL)METHYL)BENZOIC ACID
1-N-Boc-3-hydroxyazetidine
METHYL 4-((PIPERIDIN-4-YL)METHYL)BENZOATE
비스(4-아미노페닐) 에테르
D-글루코사민
2-((1-(TERT-BUTOXYCARBONYL)PIPERIDIN-4-YL)METHYL)BENZOIC ACID
2-FLUORO-5-HYDROXYBENZOIC ACID
5-FLUORO-4-HYDROXY-2-METHOXYBENZONITRILE
1,2,3-Propanetricarboxylicacid,2-hydroxy-,potassiumsalt
1-Trityl-1H-imidazole-4-carboxylic acid
N-Boc-S-Trityl-L-cysteine
2-(PIPERAZIN-1-YL)-ACETIC ACID N-(2-PHENYLETHYL)-AMIDE
TERT-BUTYL-4-(3-HYDROXYPROPYL)PIPERAZINE-1-CARBOXYLATE
4-(4-Methoxyphenyl)butyric acid
3-((1-(TERT-BUTOXYCARBONYL)PIPERIDIN-4-YL)METHYL)BENZOIC ACID
3-N-Boc-Aminomethylpiperidine
1-Boc-4-(2-formylphenyl)piperazine
Phosphatizing liquid
Disodium stannous citrate
펜타닐시트레이트
6-BROMO-2H-1,4-BENZOXAZIN-3(4H)-ONE
트리아연 디사이트레이트
Rifapentine
TERT-BUTYL 4-(4-FORMYLBENZYL)PIPERAZINE-1-CARBOXYLATE
칼륨 시트르산, 무수물
2-FLUORO-5-METHOXYBENZOIC ACID
구연산 칼슘
METHYL 2-((PIPERIDIN-4-YL)METHYL)BENZOATE
3-Amino-2-[4-butoxycarbonyl(piperazino)]pyridine
구연산 암모늄, 삼염기
아이타코닉 무수물
5-BROMO-2-FLUOROPYRIDINE-3-CARBALDEHYDE
2-Fluoropyridine-4-carboxaldehyde
구연산 나트륨
시트르산 공급 업체
글로벌( 1650)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| Hengshui Haoye Chemical Co.,Ltd. | +86-2102300 +86-18632882519 |
hy@chemcoms.com | China | 256 | 58 |
| Hefei TNJ Chemical Industry Co.,Ltd. | 0551-65418671 |
sales@tnjchem.com | CHINA | 34572 | 58 |
| Hebei Yanxi Chemical Co., Ltd. | +86-17531190177; +86-17531190177 |
peter@yan-xi.com; | China | 1016 | 58 |
| Capot Chemical Co.,Ltd. | 571-85586718 +8613336195806 |
sales@capotchem.com | China | 29811 | 60 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 |
info@tianfuchem.com | China | 21975 | 55 |
| Hangzhou FandaChem Co.,Ltd. | +8615858145714 |
fandachem@gmail.com | China | 9378 | 55 |
| Nanjing Finetech Chemical Co., Ltd. | 025-85710122 17714198479 |
sales@fine-chemtech.com | CHINA | 885 | 55 |
| Hefei TNJ Chemical Industry Co.,Ltd. | +86-0551-65418679 +86-18949832763 |
info@tnjchem.com | China | 2990 | 55 |
| Shanghai Zheyan Biotech Co., Ltd. | 18017610038 |
zheyansh@163.com | CHINA | 3620 | 58 |
| career henan chemical co | +86-0371-86658258 |
sales@coreychem.com | China | 29922 | 58 |