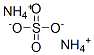황산암모늄
|
|
황산암모늄 속성
- 녹는점
- >280 °C (dec.) (lit.)
- 밀도
- 1.77 g/mL at 25 °C (lit.)
- 증기압
- <1 Pa (25 °C)
- 굴절률
- n
20/D 1.396
- 인화점
- 26 °C
- 저장 조건
- room temp
- 용해도
- H2O: 1 M at 20 °C, clear, colorless
- 물리적 상태
- Solid
- Specific Gravity
- 1.769
- 색상
- Yellow to orange
- 냄새
- Slight odor of ammonia
- pH 범위
- 5 - 6
- 수소이온지수(pH)
- 5.0-6.0 (25℃, 1M in H2O)
- 수용성
- 77 g/100 mL (25 ºC)
- 최대 파장(λmax)
- λ: 260 nm Amax: ≤0.037
λ: 280 nm Amax: ≤0.030
- Merck
- 14,555
- 안정성
- Stable. Contact with strong oxidizers may cause fire or explosion. Incompatible with strong bases.
- InChIKey
- BFNBIHQBYMNNAN-UHFFFAOYSA-N
- LogP
- -5.1 at 25℃
- CAS 데이터베이스
- 7783-20-2(CAS DataBase Reference)
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | Xi,Xn | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 10-36/37/38-22 | ||
| 안전지침서 | 37/39-26-36-24/25 | ||
| WGK 독일 | 1 | ||
| RTECS 번호 | BS4500000 | ||
| TSCA | Yes | ||
| HS 번호 | 31022100 | ||
| 유해 물질 데이터 | 7783-20-2(Hazardous Substances Data) | ||
| 독성 | LD50 orally in Rabbit: 2840 mg/kg | ||
| 기존화학 물질 | KE-01743 |
황산암모늄 C화학적 특성, 용도, 생산
개요
Ammonium sulfate was the first nitrogenous fertilizer made by the Haber-Bosch process, produced by the reaction of ammonia with sulfuric acid. In contrast with the nitrate salt, it is chemically stable, not highly hygroscopic. It also supplies supplemental sulfur to soils that may be deficient in this element, but this is of minor value when it is used on soils receiving applications of ordinary superphosphate.The disadvantages of the material are its relatively low nitrogen content, which increases storage and transportation costs, and its marked tendency to cause soil acidification, which is greater than that of any other nitrogen fertilizer material.
화학적 성질
White crystalline powder물리적 성질
White crystalline solid; orthorhombic crystal; density 1.769 g/cm3 at 20°C; melts between 511 to 515°C (in a closed system): however, in an open system, it melts with decomposition at 280°C; readily dissolves in water (solubility, 70.6 g and 104 g per 100 g water at 0°C and 100°C, respectively); insoluble in acetone, alcohol and ether.출처
Ammonium sulfate occurs in trace concentrations in the upper atmosphere. It is widely used as a fertilizer for rice and other crops. It is a source of sulfur for the soil. It is also used as an additive to supply nutrient nitrogen in fermentation processes (e.g., yeast production from molasses). It also is used for fireproofing timber and plastics, and in treatment of hides, and leather production.용도
Ammonium Sulfate is a dough conditioner, firming agent, and pro- cessing aid which is readily soluble in water with a solubility of approximately 70 g in 100 g of water at 0°c. the ph of a 0.1 molar solution in water is approximately 5.5. it is used in caramel produc- tion and as a source of nitrogen for yeast fermentation. in bakery products, up to 0.25 part per 100 parts by weight of flour is used.정의
ammonium sulphate: A whiterhombic solid, (NH4)2SO4; r.d. 1.77;decomposes at 235°C. It is very solublein water and insoluble in ethanol.It occurs naturally as the mineralmascagnite. Ammonium sulphatewas formerly manufactured from the‘ammoniacal liquors’ produced duringcoal-gas manufacture but is nowproduced by the direct reaction betweenammonia gas and sulphuricacid. It is decomposed by heating torelease ammonia (and ammoniumhydrogensulphate) and eventuallywater, sulphur dioxide, and ammonia.Vast quantities of ammoniumsulphate are used as fertilizers.생산 방법
Ammonium sulfate is a high-tonnage industrial chemical, but frequently may be considered a byproduct as well as intended end-product of manufacture. A significant commercial source of (NH4)2SO4 is its creation as a byproduct in the manufacture of caprolactam, which yields several tons of the compound per ton of caprolactam made. Ammonium sulfate also is a byproduct of coke oven operations where the excess NH3 formed is neutralized with H2SO4 to form (NH4)2SO4. In the Meresburg reaction, natural or byproduct gypsum is reacted with ammonium carbonate: CaSO4·2H2O + (NH4)2CO3 CaCO3 + (NH4)2SO4 +2 H2O The product is stable, free-flowing crystals. As a fertilizer, (NH4)2SO4 has the advantage of adding sulfur to the soil as well as nitrogen. By weight, the compound contains 21% N and 24% S. Ammonium sulfate also is used in electric dry cell batteries, as a soldering liquid, as a fire retardant for fabrics and other products, and as a source of certain ammonium chemicals.일반 설명
White odorless solid. Sinks and dissolves in water.공기와 물의 반응
Dissolves in water with evolution of some heat.반응 프로필
Ammonium sulfate is acidic in aqueous solution. When a little Ammonium sulfate is added to fused potassium nitrite, a vigorous reaction occurs attended by flame [Mellor 2:702 1946-47].Safety Profile
Moderately toxic by several routes. Human systemic effects by ingestion: hypermotility, diarrhea, nausea or vomiting. See also SULFATES. Incandescent reaction on heating with potassium chlorate. Reaction with sodmm hypochlorite gves the unstable explosive nitrogen trichloride. Incompatible with (K + NH4NO3), KNO2, (NaK + NH4NO3). When heated to decomposition it emits very toxic fumes of NOx, NH3, and SOx.Purification Methods
Crystallise it twice from hot water containing 0.2% EDTA to remove metal ions, then finally from distilled water. Dry it in a desiccator for 2 weeks over Mg(ClO4)2. After 3 recrystallisations, ACS grade had Ti, K, Fe, Na at 11, 4.4, 4.4, 3.2 ppm respectively.황산암모늄 준비 용품 및 원자재
원자재
준비 용품
Thymosin α1
Compound fertilizer
알루미늄 칼륨 황산염, 도데카수화물
리파아제
네오마이신 황산
알돌라아제, 프럭토스 이인산염
PHASEOLUS VULGARIS AGGLUTININ
Nitrogen phosphorus potassium mixed fertilizer
L-(+)-아이소루신
리소자임
Ethyl 3-bromo-2-(bromomethyl)propionate
Polyinosinic acid-polycytidylic acid
PKC-GAMMA
2-AMINO-3-CYANO-4-CHLORO-5-FORMYLTHIOPHENE
Aminoacylase
리보헥산 A
페로시안화 제2철
Chymotrypsin
알파-아밀라아제
Lincomycin hydrochloride monohydrate
L-라이신1염산염
황산 암모늄 제1철
히알루로니다제
Bromelain
제1인산암모늄
Zirconium dicarbonate
암모늄명반
라이신
피로시다제
Chorionic Gonadotropin
황산니켈암모늄
알루미늄 암모늄 황산염
3-Bromo-2-(bromomethyl)propionic acid
페니실린 감마-일라디산염 세포
카타라세
(S)-(+)-2-HYDROXY-3-METHYLBUTYRIC ACID
알코올 탈수소효소
EC 2.7.1.40
암모늄 철 황산염
diammonium magnesium bis(sulphate)
황산암모늄 공급 업체
글로벌( 691)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| Jiangsu Kolod Food Ingredients Co.,Ltd. | +86-518-85110578 +8618805133257 |
sales3257@jskolod.com | China | 130 | 60 |
| Henan Bao Enluo International TradeCo.,LTD | +86-17331933971 +86-17331933971 |
deasea125996@gmail.com | China | 2180 | 58 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 |
info@tianfuchem.com | China | 21975 | 55 |
| Hangzhou FandaChem Co.,Ltd. | +8615858145714 |
fandachem@gmail.com | China | 9378 | 55 |
| Hefei TNJ Chemical Industry Co.,Ltd. | +86-0551-65418679 +86-18949832763 |
info@tnjchem.com | China | 2990 | 55 |
| Yancheng Green Chemicals Co.,Ltd | +undefined-86-25-86655873 |
info@royal-chem.com | China | 106 | 58 |
| Shanxi Naipu Import and Export Co.,Ltd | +86-13734021967 +8613734021967 |
kaia@neputrading.com | China | 1011 | 58 |
| Shanghai Zheyan Biotech Co., Ltd. | 18017610038 |
zheyansh@163.com | CHINA | 3620 | 58 |
| career henan chemical co | +86-0371-86658258 |
sales@coreychem.com | China | 29922 | 58 |
| Hubei Jusheng Technology Co.,Ltd. | 18871490254 |
linda@hubeijusheng.com | CHINA | 28184 | 58 |