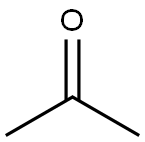
아세톤
|
|
아세톤 속성
- 녹는점
- -94 °C(lit.)
- 끓는 점
- 56 °C760 mm Hg(lit.)
- 밀도
- 0.791 g/mL at 25 °C(lit.)
- 증기 밀도
- 2 (vs air)
- 증기압
- 184 mm Hg ( 20 °C)
- 굴절률
- n
20/D 1.359(lit.)
- FEMA
- 3326 | ACETONE
- 인화점
- 1 °F
- 저장 조건
- Store at +5°C to +30°C.
- 용해도
- Miscible with water and with ethanol (96 per cent).
- 산도 계수 (pKa)
- 19.3(at 25℃)
- 물리적 상태
- Liquid
- 색상
- Colorless, invisible vapor
- Specific Gravity
- 0.79 (25/25℃)
- 냄새
- Characteristic pungent odor detectable at 33 to 700 ppm (mean = 130 ppm)
- 수소이온지수(pH)
- 5-6 (395g/l, H2O, 20°C)
- 상대극성
- 0.355
- ?? ??
- solvent
- Odor Threshold
- 42ppm
- 폭발한계
- 2.6-12.8%(V)
- 수용성
- soluble
- Merck
- 14,66
- JECFA Number
- 139
- BRN
- 63580
- Henry's Law Constant
- 2.27 at 14.9 °C, 3.03 at 25 °C, 7.69 at 35.1 °C, 11.76 at 44.9 °C (Betterton, 1991)
- 노출 한도
- TLV-TWA 1780 mg/m3 (750 ppm), STEL 2375 mg/m3 (ACGIH); 10 h–TWA 590 mg/m3 (250 ppm); IDLH 20,000 ppm (NIOSH).
- LogP
- -0.160
- CAS 데이터베이스
- 67-64-1(CAS DataBase Reference)
- NIST
- Acetone(67-64-1)
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | F,Xi,T | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 11-36-66-67-39/23/24/25-23/24/25 | ||
| 안전지침서 | 9-16-26-45-36/37-7 | ||
| 유엔번호(UN No.) | UN 1090 3/PG 2 | ||
| WGK 독일 | 3 | ||
| RTECS 번호 | AL3150000 | ||
| F 고인화성물질 | 3-10 | ||
| 자연 발화 온도 | 465 °C | ||
| TSCA | Yes | ||
| 위험 등급 | 3 | ||
| 포장분류 | II | ||
| HS 번호 | 29141100 | ||
| 유해 물질 데이터 | 67-64-1(Hazardous Substances Data) | ||
| 독성 | LD50 in rats: 10.7 ml/kg orally (Smyth) | ||
| IDLA | 2,500 ppm | ||
| 기존화학 물질 | KE-29367 |
아세톤 C화학적 특성, 용도, 생산
개요
Acetone is a flammable, colorless liquid with a pleasant odor. It is used widely as an organic solvent and in the chemical industry. It is the simplest ketone, which also goes by the name dimethyl ketone (DMK). Acetone was originally referred to as pyroacetic spirit because it was obtained from the destructive distillation of acetates and acetic acid.화학적 성질
Acetone, CH3COCH3, also known as 2-propanone and dimethylketone, is a colorless, volatile,flammable liquid that boils at 56°C (133 OF). It is misciblewith water and is oftenused as a solventin the manufacture of lacquers and paints.물리적 성질
Clear, colorless, liquid with a sweet, fragrant odor. Sweetish taste. Odor threshold concentrations ranged from 42 ppmv (Nagata and Takeuchi, 1990) to 100 ppmv (Leonardos et al., 1969). Experimentally determined detection and recognition odor threshold concentrations were 48 mg/m3 (20 ppmv) and 78 mg/m3 (33 ppmv), respectively (Hellman and Small, 1974).출처
Reported found in apple, pear, grape, pineapple, strawberry, raspberry, tomato, black currant, citrus, onion and potato; also reported found in cocoa leaves, in Mexican goosefoot and in the oils of coriander and lavender. In trace amounts it has been reportedly identified in the oil of bitter orange, in distilled wine and in coffee aroma역사
The traditional method of producing acetone in the 19th century and the beginning of the 20th century was to distill acetates, particularly calcium acetate, Ca(C2H3O2)2.Weizmann discovered a process to produce butyl alcohol and acetone from the bacterium Clostridium acetobutylicum in 1914. With England’s urgent demand for acetone, Winston Churchill (1874–1965) enlisted Weizmann to develop the Weizmann process for acetone production on an industrial scale. Fermentation and distillation techniques for acetone production were replaced starting in the 1950s with the cumene oxidation process . In this process, cumene is oxidized to cumene hydroperoxide, which is then decomposed using acid to acetone and phenol. This is the primary method used to produce phenol, and acetone is produced as a co-product in the process, with a yield of about 0.6:1 of acetone to phenol.
용도
Acetone′s luminesence intensity is dependent upon the solution components 1. The absorption of UV light by acetone, results in its photolysis and the production of radials 1.정의
ChEBI: A methyl ketone that consists of propane bearing an oxo group at C2.생산 방법
Acetone is obtained by fermentation as a by-product of n-butyl alcohol manufacture, or by chemical synthesis from isopropyl alcohol; from cumene as a by-product in phenol manufacture; or from propane as a by-product of oxidation-cracking.일반 설명
A clear colorless liquid with a sweetish odor. Flash point 0°F. Less dense than water. Vapors are heavier than air. Used as a solvent in paint and nail polish removers.공기와 물의 반응
Highly flammable. Water soluble.반응 프로필
Acetone was reported that a mixture of Acetone and chloroform, in a residue bottle, exploded. Since addition of Acetone to chloroform in the presence of base will result in a highly exothermic reaction, Acetone is thought that a base was in the bottle [MCA Case History 1661. 1970]. Also, Nitrosyl chloride, sealed in a tube with a residue of Acetone in the presence of platinum catalyst, gave an explosive reaction [Chem. Eng. News 35(43):60. 1967]. The reaction of nitrosyl perchlorate and Acetone ignites and explodes. Explosions occur with mixtures of nitrosyl perchlorate and primary amine [Ann. Chem. 42:2031. 1909]. Reacts violently with nitric acid. Also causes exothermic reaction when in contact with aldehydes.건강위험
The acute toxicity of acetone is low. Acetone is primarily a central nervous system depressant at high concentrations (greater than 12,000 ppm). Unacclimated volunteers exposed to 500 ppm acetone experienced eye and nasal irritation, but it has been reported that 1000 ppm for an 8-hour day produced no effects other than slight transient irritation to eyes, nose, and throat. Therefore there are good warning properties for those unaccustomed to working with acetone; however, frequent use of acetone seems to cause accommodation to its slight irritating properties. Acetone is practically nontoxic by ingestion. A case of a man swallowing 200 mL of acetone resulted in his becoming stuporous after 1 hour and then comatose; he regained consciousness 12 hour later. Acetone is slightly irritating to the skin, and prolonged contact may cause dermatitis. Liquid acetone produces moderate transient eye irritation. Acetone has not been found to be carcinogenic in animal tests or to have effects on reproduction or fertility.화재위험
HIGHLY FLAMMABLE: Will be easily ignited by heat, sparks or flames. Vapors may form explosive mixtures with air. Vapors may travel to source of ignition and flash back. Most vapors are heavier than air. They will spread along ground and collect in low or confined areas (sewers, basements, tanks). Vapor explosion hazard indoors, outdoors or in sewers. Runoff to sewer may create fire or explosion hazard. Containers may explode when heated. Many liquids are lighter than water.인화성 및 폭발성
Acetone is extremely flammable (NFPA rating = 3), and its vapor can travel a considerable distance to an ignition source and "flash back." Acetone vapor forms explosive mixtures with air at concentrations of 2 to 13% (by volume). Carbon dioxide or dry chemical extinguishers should be used for acetone fires.Pharmaceutical Applications
Acetone is used as a solvent or cosolvent in topical preparations, and as an aid in wet granulation.It has also been used when formulating tablets with water-sensitive active ingredients, or to solvate poorly water-soluble binders in a wet granulation process. Acetone has also been used in the formulation of microspheres to enhance drug release.Owing to its low boiling point, acetone has been used to extract thermolabile substances from crude drugs.공업 용도
Acetone is valuable solvent component in acrylic/nitrocellulose automotive lacquers. Acetone is the solvent of choice in film coatings operations which use vinylidene chloride-acrylonitrile copolymer formulations.Other ketones that may be used in these film coating operations include methyl isobutyl ketone, ethyl n-amyl ketone, and diisobutyl ketone.Acetone, blends of MIBK and MEK, methyl namyl ketone, ethyl n-amyl ketone, and diisobutyl ketone are all useful solvents for vinyl resin copolymers. The presence of one of the slower evaporating ketones in the solvent blend prevents quick drying, improves flow, and gives blush resistance to the coating. Acetone is also used as a resin thinner in polyester resins and as a clean up solvent for the resin reactor kettle.In solvents industry, Acetone is a component of solvent blends in urethane, nitrile rubber, and neoprene industrial adhesives.Acetone is the primary solvent in resin-type adhesives and pressure sensitive chlorinated rubber adhesives. Acetone also can be used to extract fats, oils, waxes, and resins from natural products, to dewax lubricating oils, and to extract certain essential oils.Acetone is also an important chemical intermediate in the preparation of several oxygenated solvents including the ketones, diacetone alcohol, mesityl oxide, methyl isobutyl ketone, and isophorone.Safety
Acetone is considered moderately toxic, and is a skin irritant and severe eye irritant. Skin irritation has been reported due to its defatting action, and prolonged inhalation may result in headaches. Inhalation of acetone can produce systemic effects such as conjunctival irritation, respiratory system effects, nausea, and vomiting.LD50 (mouse, oral): 3.0 g/kg
LD50 (mouse, IP): 1.297 g/kg
LD50 (rabbit, oral): 5.340 g/kg
LD50 (rabbit, skin): 0.2 g/kg
LD50 (rat, IV): 5.5 g/kg
LD50 (rat, oral): 5.8 g/kg
잠재적 노출
It is used as a solvent in nail polish remover and many other chemicals. Used in the production of lubricating oils and as an intermediate in the manufacture of chloroform and of various pharmaceuticals and pesticides.Carcinogenicity
Acetone may be weakly genotoxic, but the majority of assays were negative.7 It was not tumorigenic in skin painting studies in mice.The 2003 ACGIH threshold limit valuetime- weighted average (TLV-TWA) for acetone is 750ppm (1780mg/m3) with a short-term excursion level of 1000ppm (2380mg/m3).
환경귀착
Biological. Following a lag time of 20 to 25 h, acetone degraded in activated sludge (30 mg/L) at a rate constant ranging from 0.016 to 0.020/h (Urano and Kato, 1986). Soil bacteria can mineralize acetone to carbon dioxide (Taylor et al., 1980). Bridié et al. (1979) reported BOD and COD values of 1.85 and 1.92 g/g using filtered effluent from a biological sanitary waste treatment plant. These values were determined using a standard dilution method at 20 °C for a period of 5 d. Similarly, Heukelekian and Rand (1955) reported a 5-d BOD value of 0.85 g/g which is 38.5% of the ThOD value of 2.52 g/g. Waggy et al. (1994) reported 5, 15, and 28-d BOD values of 14, 74, and 74%, respectively. Using the BOD technique to measure biodegradation, the mean 5-d BOD value (mM BOD/mM acetone) and ThOD were 1.52 and 38.0%, respectively (Vaishnav et al., 1987).Photolytic. Photolysis of acetone in air yields carbon monoxide and free radicals, but in isopropanol, pinacol is formed (Calvert and Pitts, 1966). Photolysis of acetone vapor with nitrogen dioxide via a mercury lamp gave peroxyacetyl nitrate as the major product with smaller quantities of methyl nitrate (Warneck and Zerbach, 1992).
Chemical/Physical. Hypochlorite ions, formed by the chlorination of water for disinfection purposes, may react with acetone to form chloroform. This reaction is expected to be significant within the pH range of 6 to 7 (Stevens et al., 1976).
저장
Acetone should be used only in areas free of ignition sources, and quantities greater than 1 liter should be stored in tightly sealed metal containers in areas separate from oxidizers.운송 방법
UN1090 Acetone, Hazard Class: 3; Labels: 3-Flammable liquid비 호환성
Acetone reacts violently with oxidizing agents, chlorinated solvents, and alkali mixtures. It reacts vigorously with sulfur dichloride, potassium t-butoxide, and hexachloromelamine. Acetone should not be used as a solvent for iodine, as it forms a volatile compound that is extremely irritating to the eyes.폐기물 처리
Consult with environmental regulatory agencies for guidance on acceptable disposal practices. Generators of waste containing this contaminant (≥100 kg/mo) must conform with EPA regulations governing storage, transportation, treatment, and waste disposal. Incineration.Regulatory Status
Included in the FDA Inactive Ingredients Database (inhalation solution; oral tablets; topical preparations). Included in the Canadian List of Acceptable Non-medicinal Ingredients. Included in nonparenteral medicines licensed in the UK.아세톤 준비 용품 및 원자재
원자재
큐멘
2-Amino-3-chlorobenzoic acid
프로필렌
Waste molasses
tert-부틸알코올
고무시멘트 희석제
크롬산 아연
벤젠
큐멘하이드로퍼옥사이드
디이소프로필벤젠
Lng
아세트산 칼슘
파라시멘
상수리나무목초액추출물
준비 용품
PYRIDINE-2-SULFONIC ACID
Chalcone
Pyridazin-3-amine
올시프레나린
2-(4-BENZYLOXYPHENYL)ETHANOL
PROPIONAMIDINE
CHEMBRDG-BB 6088138
9,9-dimethylcarbazine
3-(-Hydroxyphenethyl)-2-imino thiazolidine
2-(4-(CHLOROMETHYL)THIAZOL-2-YL)GUANIDINE
1-Phenylethanone phenylhydrazone
7-Methoxycoumarin
사질산 펜타에리트리톨
7-Ethoxycoumarin
2-요오드아세트아미드
3,3',4,4'-벤조페논 테트라카본산 2무수물
에톡시퀸
4-Hydroxybenzylideneacetone
Dimethyl acetylmethylphosphonate
1,1,3,3-TETRABROMOACETONE
Amikacin sulfate salt
요오드포름
4-CARBOXYPHENYL ISOTHIOCYANATE
아미카신 황산염
아이소포론
5-METHOXY-2-METHYLINDOLE
3,4-DIHYDRO-2H-1,5-BENZODIOXEPINE-7-CARBALDEHYDE
4-(2-THIENYL)BUT-3-EN-2-ONE
에핀에피린
H-GLU-LYS-OH
2,5-Dichlorothiophene-3-sulfonamide
2-(MORPHOLINE-4-CARBONYL)-BENZOIC ACID
2-BROMODODECANOIC ACID
신코펜
3-메틸-1-부틴-3-올
3-DIMETHYLAMINOPROPIOPHENONE HYDROCHLORIDE
다이비닐테트라메틸다이실록산
3-Methyl-2-thiophenecarboxylic acid
methyl 3,4-dihydro-4-oxoquinazoline-7-carboxylate
7-ALLYLOXYCOUMARIN
아세톤 공급 업체
글로벌( 0)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|
아세톤 관련 검색:
펜타논-2 4'-히드록시프로피오페논 디스프로슘 트리(2,2,6,6-테트라메틸-3,5-헵탄디오네이트) 프라세오디뮴 트리(디피발로일메탄) 트리(2,2,6,6-테트라메틸-3,5-헵타네디오나토)이테르븀 아세톤 프로피오페논 시트르산(구연산) 2,4-펜탄디온 디아세톤알코올 피루브산
TERT-BUTYL ISOCYANIDE
TRIS(2,2,6,6-TETRAMETHYL-3,5-HEPTANEDIONATO)EUROPIUM(III)
PHENYLSELENOL
LANTHANUM ACETYLACETONATE
4-Methoxyphenylacetone
2-Bromo-4'-fluoroacetophenone
Diacetone-D-glucose