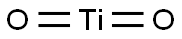이산화티타늄
|
|
이산화티타늄 속성
- 녹는점
- 1840 °C
- 끓는 점
- 2900 °C
- 밀도
- 4.26 g/mL at 25 °C(lit.)
- 굴절률
- 2.61
- 인화점
- 2500-3000°C
- 저장 조건
- Store at +5°C to +30°C.
- 용해도
- Practically insoluble in water. It does not dissolve in dilute mineral acids but dissolves slowly in hot concentrated sulfuric acid.
- 물리적 상태
- powder
- Specific Gravity
- 4.26
- 색상
- White to slightly yellow
- 수소이온지수(pH)
- 7-8 (100g/l, H2O, 20℃)(slurry)
- 냄새
- at 100.00?%. odorless
- 수용성
- insoluble
- Crystal Structure
- Orthorhombic, Pcab
- Merck
- 14,9472
- 노출 한도
- ACGIH: TWA 10 mg/m3
OSHA: TWA 15 mg/m3
NIOSH: IDLH 5000 mg/m3; TWA 2.4 mg/m3; TWA 0.3 mg/m3
- CAS 데이터베이스
- 13463-67-7(CAS DataBase Reference)
- IARC
- 2B (Vol. 47, 93) 2010
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | |||
|---|---|---|---|
| 위험 카페고리 넘버 | 10-20-22 | ||
| 안전지침서 | 26-36-25-2-36/37-45-36/37/39 | ||
| WGK 독일 | - | ||
| HS 번호 | 28230000 | ||
| 유해 물질 데이터 | 13463-67-7(Hazardous Substances Data) | ||
| 독성 | LD50 orally in Rabbit: > 10000 mg/kg | ||
| IDLA | 5,000 mg/m3 | ||
| 기존화학 물질 | KE-33900 |
이산화티타늄 C화학적 특성, 용도, 생산
개요
Titanium dioxide, TiO2, is a white powder and has the greatest hiding power of all white pigments. It is noncombustible; however, it is a powder and, when suspended in air, may cause a dust explosion if an ignition source is present. It is not listed in the DOT Hazardous Materials Table, and the DOT does not consider it hazardous in transportation. The primary uses are as a white pigment in paints, paper, rubber, and plastics; in cosmetics; in welding rods; and in radioactive decontamination of the skin.화학적 성질
Ttitanium dioxide is an odorless white powder.물리적 성질
The naturally occurring dioxide exists in three crystal forms: anatase, rutile and brookite. While rutile, the most common form, has an octahedral structure. Anatase and brookite have very distorted octahedra of oxygen atoms surrounding each titanium atom. In such distorted octahedral structures, two oxygen atoms are relatively closer to titanium than the other four oxygen atoms. Anatase is more stable than the rutile form by about 8 to 12 kJ/mol (Cotton, F.A., Wilkinson, G., Murillo, C.A and M Bochmann. 1999. Advanced Inorganic Chemistry, 6th ed, p. 697, New York: John Wiley & Sons) Other physical properties are: density 4.23g/cm3; Mohs hardness 5.8 g/cm3 ( anatase and brookite) and 6.2 g/cm3 ( rutile); index of refraction 2.488 (anatase), 2.583 (brookite) and 2.609 (rutile); melts at 1,843°C; insoluble in water and dilute acids; soluble in concentrated acids.용도
Titanium dioxide is an extreme white and bright compound with high index of refraction. In paints it is a white pigment and an opacifying agent.It is in house paints, water paints, lacquers, enamels, paper filling and coating, rubber, plastics, printing ink, synthetic fabrics, floor coverings, and shoe whiteners. Also, it is used in colorants for ceramics and coatings for welding rods. A rutile form of the dioxide is used in synthetic gem stones.생산 방법
Titanium dioxide occurs naturally as the minerals rutile (tetragonal structure), anatase (tetragonal structure), and brookite (orthorhombic structure).Titanium dioxide may be prepared commercially by either the sulfate or chloride process. In the sulfate process a titanium containing ore, such as ilemenite, is digested in sulfuric acid. This step is followed by dissolving the sulfates in water, then precipitating the hydrous titanium dioxide using hydrolysis. Finally, the product is calcinated at high temperature. In the chloride process, the dry ore is chlorinated at high temperature to form titanium tetrachloride, which is subsequently oxidized to form titanium dioxide.
제조 방법
Titanium dioxide is mined from natural deposits. It also is produced from other titanium minerals or prepared in the laboratory. Pigment-grade dioxide is produced from the minerals, rutile and ilmenite. Rutile is converted to pigment grade rutile by chlorination to give titanium tetrachloride, TiCl4. Anhydrous tetrachloride is converted back to purified rutile form by vapor phase oxidation.Anatase form is obtained by hydrolytic precipitation of titanium(IV) sulfate on heating. The mineral ilmenite is treated with concentrated sulfuric acid. Heating the sulfate solution precipitates hydrous titanium oxide. The precipitate is calcined to expel all water.
Titanium dioxide also can be prepared by heating Ti metal in air or oxygen at elevated temperatures.
일반 설명
Two main physico-chemically distinct polymorphs of TiO2 are anatase and rutile. Anatase has a higher photocatalytic activity than rutile but is thermodynamically less stable.위험도
Lower respiratory tract irritant. Possible carcinogen.건강위험
Titanium dioxide is a mild pulmonary irritant and is generally regarded as a nuisance dust.Pharmaceutical Applications
Titanium dioxide is widely used in confectionery, cosmetics, and foods, in the plastics industry, and in topical and oral pharmaceutical formulations as a white pigment.Owing to its high refractive index, titanium dioxide has lightscattering properties that may be exploited in its use as a white pigment and opacifier. The range of light that is scattered can be altered by varying the particle size of the titanium dioxide powder. For example, titanium dioxide with an average particle size of 230nm scatters visible light, while titanium dioxide with an average particle size of 60nm scatters ultraviolet light and reflects visible light.
In pharmaceutical formulations, titanium dioxide is used as a white pigment in film-coating suspensions, sugar-coated tablets, and gelatin capsules. Titanium dioxide may also be admixed with other pigments.
Titanium dioxide is also used in dermatological preparations and cosmetics, such as sunscreens.
Safety Profile
A nuisance dust. A human skin irritant. Questionable carcinogen with experimental carcinogenic, neoplastigenic, and tumorigenic data. Violent or incandescent reaction with metals at high temperatures (e.g., aluminum, calcium, magnesium, potassium, sodium, zinc, lithium). See also TITANIUM COMPOUNDS.Safety
Titanium dioxide is widely used in foods and oral and topical pharmaceutical formulations. It is generally regarded as an essentially nonirritant and nontoxic excipient.잠재적 노출
Titanium dioxide is a white pigment used as a pigment in paint; in the rubber, plastics, ceramics, paint, and varnish industries, in dermatological preparations; and is used as a starting material for other titanium compounds; as a gem; in curing concrete; and in coatings for welding rods. It is also used in paper and cardboard manufacture.Carcinogenicity
Carcinogenesis. In a 1985 study, rats (CD) were exposed to graded airborne concentrations (0, 10, 50, and 250mg/m3) of TiO2 6 h/day, 5 days/week, for 2 years. The majority of the particles were in the respirable range (84% ≤13 mmMMD). All responses were confined to the lungs. At the lowest dose, the histopathological evaluation of the lungs revealed dust-laden macrophages in the alveolar ducts and adjacent alveoli with pneumocyte hyperplasia. At the two highest concentrations, there were increases in lung weight, accumulation of dust in the macrophages, foamy macrophage responses, type II pneumocyte hyperplasia, alveolar proteinosis, alveolar bronchiolization, cholesterol granulomas, focal pleurisy, and dust deposition in the tracheobronchiolar lymph nodes. At the 250mg/m3 exposure concentration, bronchiole alveolar adenomas (males: control 2/79, 250mg/m3 12/79; females: control 0/79, 250mg/m3 13/79) increased. Additionally, 13/79 females at the 250mg/m3 dose showed squamous cell carcinoma, compared with none in 79 controls. Theauthorsnoted that this responsemight have little biological relevance to humans because of the overload of respiratory clearance mechanisms and also pointed out that the type, location, and development of the tumors were different from those in human lung tumors. It is not clear that the nasal cavity epithelium was examined. However, the nasal cavity load would be expected to be higher in the rats because of anatomic structure, whereas the lung deposition should be higher in humans because we are, in part, mouth breathers.저장
Titanium dioxide is extremely stable at high temperatures. This is due to the strong bond between the tetravalent titanium ion and the bivalent oxygen ions. However, titanium dioxide can lose small, unweighable amounts of oxygen by interaction with radiant energy. This oxygen can easily recombine again as a part of a reversible photochemical reaction, particularly if there is no oxidizable material available. These small oxygen losses are important because they can cause significant changes in the optical and electrical properties of the pigment.Titanium dioxide should be stored in a well-closed container, protected from light, in a cool, dry place.
비 호환성
Titanium dioxide is incompatible with strong oxidizers and strong acids. Violent or incandescent reactions may occur with metals (e.g., aluminum, calcium, magnesium, potassium, sodium, zinc, and lithium).폐기물 처리
Land fill.Regulatory Status
Accepted as a food additive in Europe. Included in the FDA Inactive Ingredients Database (dental paste; intrauterine suppositories; ophthalmic preparations; oral capsules, suspensions, tablets; topical and transdermal preparations). Included in nonparenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.이산화티타늄 준비 용품 및 원자재
원자재
Iron ore
(S)-tert-Butyl 6-amino-2-((tert-butoxycarbonyl)amino)hexanoate hydrochloride
티타늄
Iron filings
삼산화안티모니
산화알루미늄
인산
Metatitanic acid
High titanium slag
탄산 칼륨
Petroleum coke
산소, 냉각된 액체
Concentrated sulfuric acid
알루미늄 클로로수화물
금홍석
TITANIUM OXYSULFATE
수산화나트륨
철분
코크스
황산제일철7수화물
ILMENITE
황산
준비 용품
Various color alkyd ready mixed paint
Styrene-acrylic latex paint for exterior wall
Alkyd resin paint
Exterior wall coating
유황
Interior and exterior wall coating
디칼륨 헥사플루오로티타늄산염
latex paint
Master-batches
Iron oxide red alkyd primer
Acrylic latex paint for interior wall
Acrylic latex paint for exterior wall
Latex paint for interior and exterior wall
Red lead alkyd antirust paint
Polyester resin paint
Alkyd enamel
PAINT
Anticorrosive coating
Interior wall coating
Amino resin paint
Epoxy polyester powder coating,mixed type
Thermosetting powder coating
이산화티타늄 공급 업체
글로벌( 533)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| AS WATER CO., LTD. | +86-18994963526 +8618994963526 |
1346073549@qq.com | United Kingdom | 173 | 58 |
| Hebei Dangtong Import and export Co LTD | +8615632909969 |
admin@hbdangtong.com | China | 858 | 58 |
| Henan Bao Enluo International TradeCo.,LTD | +86-17331933971 +86-17331933971 |
deasea125996@gmail.com | China | 2180 | 58 |
| Xiamen Wonderful Bio Technology Co., Ltd. | +8618759294010 |
Beata@xmwonderfulbio.com | China | 313 | 58 |
| Henan DaKen Chemical CO.,LTD. | +86-371-66670886 |
info@dakenchem.com | China | 18729 | 58 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 |
info@tianfuchem.com | China | 21975 | 55 |
| Hangzhou FandaChem Co.,Ltd. | +8615858145714 |
fandachem@gmail.com | China | 9378 | 55 |
| Hefei TNJ Chemical Industry Co.,Ltd. | +86-0551-65418679 +86-18949832763 |
info@tnjchem.com | China | 2990 | 55 |
| career henan chemical co | +86-0371-86658258 |
sales@coreychem.com | China | 29922 | 58 |
| Wuhan Boyuan Import & Export Co., LTD | +86-15175982296 |
Mike@whby-chem.com | China | 962 | 58 |